

|
Margin Optimization for Fractionated Breath-hold Radiation Therapy of Small Lung LesionsW. G. O'Dell2, M. C. Schell2, P. Okunieff |
| I. Introduction |

|
| End-expiration breath holding often enables one to reduce the radiotherapy treatment margin size for lesions <20 mm in diameter such that a lethal dose can be administered to the tumor while keeping the volume of healthy tissue receiving toxic dose within clinically acceptable limits. The primary targeting problem is the variability in target location (TLV) over repeated breath-holds that results in dose field broadening [1]. We seek an optimal treatment margin specification for any given TLV that will result in a tumoricidal dose being delivered to the target while minimizing the volume of surrounding healthy tissue that is likewise ablated. Furthermore, we hypothesize that it is possible to exploit the statistically-random nature of TLV to utilize extremely small, even negative-valued margins. |
II. Methods
III. Results
|
Optimizing Positive Margins
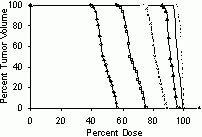
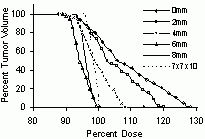
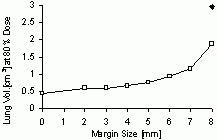
Computer-generated dataset: The optimal (positive) margin was always smaller than the corresponding TLV for TLV < 4 mm. The slope of the optimal-margin versus location-variability relationship for TLV values greater than or equal to 2 mm was 1.47, with intercept ñ1.87 mm. Lung Lesion dataset: The optimal margin size was always greater than the corresponding TLV, with a slope of 1.6 and intercept of 0.713. Optimal variability values could not be determined for margins <2.5 mm because the minimum tumor dose was already less than the desired minimum 90% dose level. Shifting the lung lesion curve accordingly (downward 2.5mm) gives a similar result to the simulated results. | 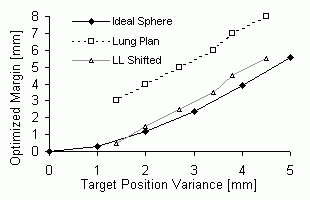
| |
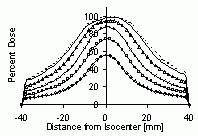
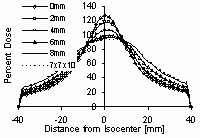
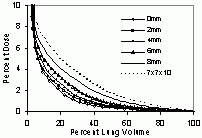
| Optimizing for Very Small (& Negative) Margins: Lung Lesion Plan, TLV of 4mm | BEFORE SCALING | AFTER DOSE AMPLIFICATION | dose profiles and DVHs | dose profiles and DVHs | surrounding lung DVH (top)
volume seeing >= 80% dose(bot) |
 |
 |
|
 |
 |
|
IV. Conclusions
| For a given target location variability (TLV) in the range of 1-4 mm (encompassing the expected physiologic range when using repeated end-expiration breath holding [2]), the optimal margin size was found to be approximately equal to the standard deviation in the target position. Since the planning treatment margin has traditionally been defined to match the target range, our results suggest that the volume of irradiated tissue can be reduced substantially. For an ideal, 12 mm-diameter spherically symmetric target and dose distribution and a TLV of 4 mm, the optimal treatment margin is 3.9 mm, resulting in 1.06 cm3 of surrounding, healthy tissue receiving >80% maximal dose ñ a drop of 85% compared with conventional margins. The technique has been extended successfully to include the use of very small and even negative-valued margins, when the proper dose amplification is applied. The use of negative margins can give another 50% or greater decrease. | ||
| ||
Acknowledgments
The Authors wish to acknowledge the support of BrainLAB GmbH, and
the American Cancer Society (RSG-02-155-01-CCE)