

|
Reproducibility of Internal Target Positions for Breath-held Conformal External-beam RadiotherapyW. G. O'Dell, C. R. Maurer, Jr., M. C. Schell, A. Sandhu, P. Okunieff |
| I. Introduction |

|
| Recent advances in Stereotactic Radiosurgery/Conformal Radiotherapy have made it possible to deliver precise radiation therapy to small lesions while preserving function to surrounding structures. Unfortunately, the application of 3D conformal radiotherapy to mobile tumors in the lung and liver is geared toward slowing the progression of disease rather than obtaining a cure. Here, the traditional therapeutic approach is to measure the range over which the tumor moves during the respiratory cycle and to then irradiate a volume that encloses the entire tumor over its entire motion range. The oncologist’s dilemma is that prescribing a lethal radiation dose to the area would not only kill the tumor but also damage a sufficiently large volume of healthy tissue to cause significant clinical repercussions, including death. Our ultimate goal is to hit, with a very focused and high-dose radiation beam, moving targets within the body with such high precision that we will cure these cancer patients of their disease while sparing the surrounding healthy tissue. |
II. Objectives
The fundamental physiologic questions relevant to this approach are:
|
III. Methods
|
1. Imaging Three sets of chest MRI volume data were acquired during periods of breath-hold (BH) at relaxed end-expiration in a patient with five ~8--15mm diameter lung lesions. The patient was coached to perform 2 deep inspiration breathing cycles followed by a relaxed exhale and hold. This same procedure was performed during the planning CT and radiotherapy treatment sessions. Each MRI 3D dataset consisted of 168 overlapping sagittal slices with slice thickness 4 mm and slice center-to-center separation of 1 mm. Breath holding for the duration of acquisition (~32s) was well-tolerated by the patient. An MRI end-inspiration BH dataset was also acquired to compute total respiratory motion.
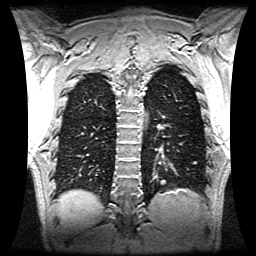
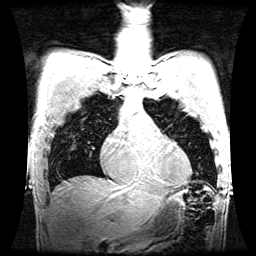
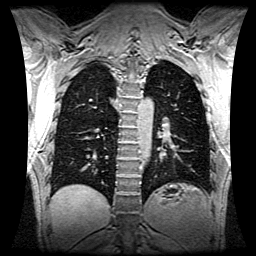
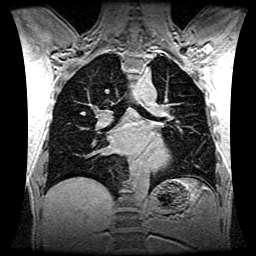
| |||||||||||||||||||||||||||||||||||
|
2. Position Variability For each of the 5 lesions, the standard deviations [mm] about the mean position (assuming a Gaussian distribution) were computed over the 3 end-expiration breath-hold trials. These values are similar to those found previously[1,2] for end-expiration breath-hold reproducibility of diaphragm position.
|
IV. Radiotherapy Planning
1. Margin Specification: 7x7x10mm [APxRLxSI]
| |||||||||||||||
2. Radiation Dose
| |||||||||||||||
|
3. Patient Repositioning using ExacTrac Automatic registration of 8 surface markers affixed to the patient’s chest and abdomen was performed using the ExacTrac system (BrainLAB, AG) incorporated into our Novalis treatment facility. The positions of all 8 skin markers were monitored in real-time in all three dimensions to determine the extent of variation in their position. A deviation of any surface marker position exceeding 2mm in any direction, using the initial planning scan locations for reference, signaled cessation of treatment until the patient repositioned himself through another relaxed exhalation. | |||||||||||||||
4. Lesion Localization Verification
| |||||||||||||||
V. Treatment Results
|
VI. Conclusions
|
The variability in target 1D position over repeated end-expiration breath-holds
was found, on average, to be 1.1—2.3mm with the largest motion in the SI direction.
Using these values as a guide, radiotherapy treatments were planned and administered
to 5 lung lesions in a single patient. 50Gy doses were given to each lesion while
maintaining a favorable dose-volume histogram for the surrounding lung tissue. We
have shown the feasibility of using breath-holding and surface marker registration
to perform fractionated external-beam radiotherapy of internal, mobile targets.
Although a curative response of these tumors was likely achieved, our future work
will focus on obtaining a larger number of trials and better statistical analysis
of tumor position variability to enable us to further limit the margin size,
decreasing the incidence of pneumonitis. Real-time motion modeling and beam
gating are also being developed.
|
Acknowledgments
The Authors wish to acknowledge the support of BrainLAB GmbH, and the University
of Rochester‘s James. P. Wilmot Cancer Center